Waa maxay titanium dioxide?
Qaybta ugu weyn ee titanium dioxide waa TIO2, kaas oo ah midab kiimiko oo aan dabiici ahayn oo qaab cad ama budo ah. Ma aha sun, wuxuu leeyahay caddaan iyo dhalaal sare, waxaana loo arkaa midabka cad ee ugu fiican ee lagu hagaajin karo cadaanta walxaha. Waxaa si weyn loogu isticmaalaa warshadaha sida dahaarka, balaastikada, caagga, warqadda, khad, dhoobada, galaaska, iwm.

Ⅰ.Jaantuska silsiladda warshadaha ee titanium dioxide:
(1)Kor u kaca silsiladda warshadaha titanium dioxide waxay ka kooban tahay walxo cayriin ah, oo ay ku jiraan ilmenite, titanium concentrate, rutile, iwm.
(2)Dhexdhexaadku wuxuu tilmaamayaa alaabada titanium dioxide.
(3) Qaybta hoose waa goobta lagu isticmaalo titanium dioxide.Titanium dioxide waxaa si weyn loogu isticmaalaa dhinacyo kala duwan sida dahaarka, balaastikada, sameynta waraaqaha, khad, cinjir, iwm.

Ⅱ. Qaab-dhismeedka kiristaalka ee titanium dioxide:
Titanium dioxide waa nooc ka mid ah isku-dhafka polymorphous, kaas oo leh saddex qaab oo kiristaal caadi ah oo dabiici ah, kuwaas oo kala ah anatase, rutile iyo brookite.
Labada nooc ee rutile iyo anatase waxay ka tirsan yihiin nidaamka kiristaalka tetragonal, kuwaas oo deggan heerkulka caadiga ah; brookite wuxuu ka tirsan yahay nidaamka kiristaalka orthorhombic, oo leh qaab-dhismeed kiristaal aan degganayn, sidaa darteed qiimo yar ayuu ku leeyahay warshadaha hadda.

Saddexda qaab-dhismeed, marxaladda rutile waa tan ugu xasilloon. Marxaladda Anatase waxay si aan dib-u-noqosho lahayn isu beddeli doontaa marxaladda rutile oo ka sarreysa 900°C, halka marxaladda brookite ay si aan dib-u-noqosho lahayn isu beddeli doonto marxaladda rutile oo ka sarreysa 650°C.
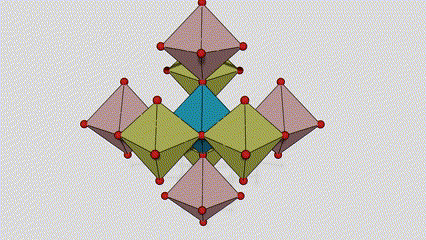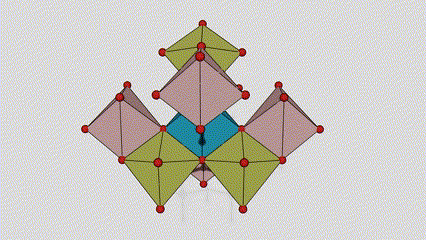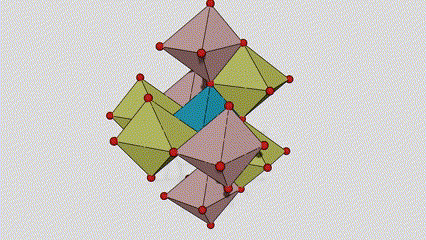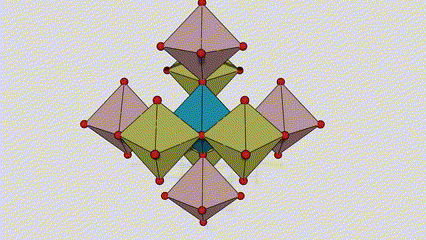
(1) Titamiin dioxide oo ah nooca Rutile phase
Marka la eego marxaladda rutile titanium dioxide, atamka Ti waxay ku yaalliin bartamaha shabagga kiristaalka, lix atamka oksijiintana waxay ku yaalliin geesaha octahedron-ka titanium-oxygen. Octahedron kasta wuxuu ku xiran yahay 10 octahedron oo ku wareegsan (oo ay ku jiraan siddeed gees oo wadaag ah iyo laba gees oo wadaag ah), laba molecules TiO2-na waxay sameeyaan unug unug.
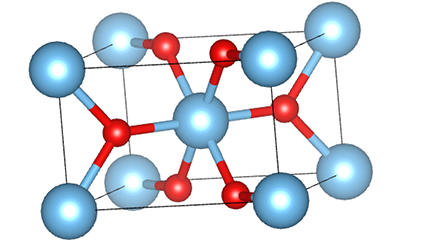
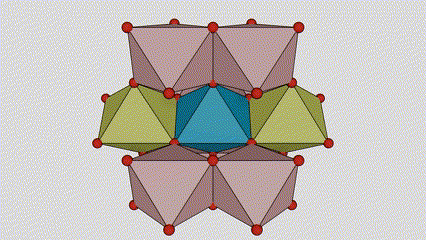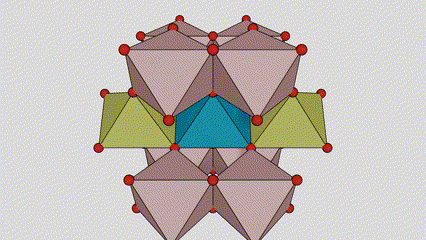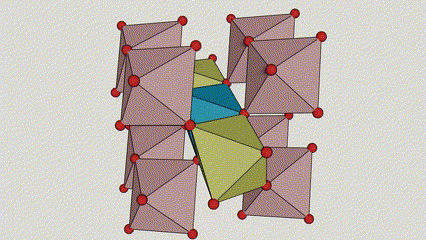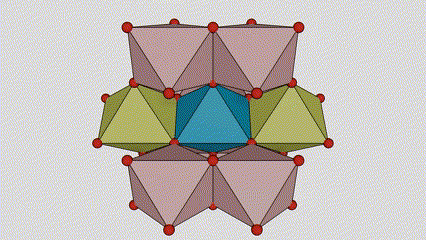
Jaantuska jaantuska ee unugga kiristaalka ee marxaladda rutile titanium dioxide (bidix)
Habka isku xirka ee titanium oxide octahedron (midig)
(2) Titamiin dioxide oo ah wejiga Anatase
Marxaladda anatase titanium dioxide, octahedron kasta oo titanium-oxygen ah wuxuu ku xiran yahay 8 octahedron oo ku wareegsan (4 gees oo wadaag ah iyo 4 gees oo wadaag ah), 4 molecules TiO2-na waxay sameeyaan unug unug.
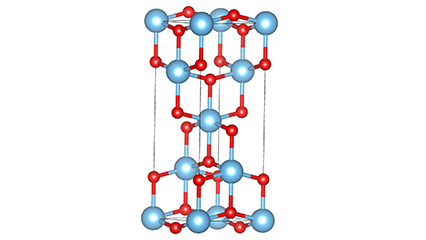
Jaantuska jaantuska ee unugga kiristaalka ee marxaladda rutile titanium dioxide (bidix)
Habka isku xirka ee titanium oxide octahedron (midig)
Ⅲ. Hababka Diyaarinta Titanium Dioxide:
Habka wax soo saarka ee titanium dioxide waxaa inta badan ku jira habka sulfuric acid iyo habka koloriinaynta.

(1) Habka aashitada sulfuric
Habka aashitada sulfuric ee soo saarista titanium dioxide waxay ku lug leedahay falgalka aashitada ee budada birta titanium oo leh aashitada sulfuric ee xooggan si loo soo saaro titanium sulfate, kaas oo markaa la hawo geliyo si loo soo saaro aashitada metatitanic. Ka dib kalsiyeynta iyo burburinta, waxaa la helaa alaabada titanium dioxide. Habkani wuxuu soo saari karaa anatase iyo rutile titanium dioxide.
(2) Habka koloriinta
Habka koloriin-ku-darka ee wax soo saarka titanium dioxide waxaa ku jira isku darka budada rutile ama titanium-ka sare leh ee kookaha ka dibna la sameeyo koloriin-ku-darka heerkulka sare leh si loo soo saaro titanium tetrachloride. Ka dib oksaydheynta heerkulka sare, badeecada titanium dioxide waxaa laga helaa shaandhaynta, dhaqidda biyaha, qalajinta, iyo burburinta. Habka koloriin-ku-darka ee wax soo saarka titanium dioxide wuxuu soo saari karaa oo keliya alaabada rutile.
Sidee loo kala saaraa saxnaanta titanium dioxide?
I. Hababka Jirka:
(1)Habka ugu fudud waa in la isbarbardhigo qaab-dhismeedka adigoo taabanaya. Titanium dioxide been abuur ah wuxuu dareemaa mid siman, halka titanium dioxide-ka dhabta ah uu dareemo mid qallafsan.

(2)Markaad biyo ku raaciso, haddii aad gacantaada saarto qaar ka mid ah titanium dioxide, kan been abuurka ah si fudud ayaa loo dhaqi karaa, halka kan dhabta ah uusan si fudud u dhaqi karin.

(3)Koob biyo nadiif ah qaado oo ku rid titanium dioxide. Kan dul sabeynaya waa mid dhab ah, halka kan salka ku degaya uu yahay mid been abuur ah (habkani waxaa laga yaabaa inaanu u shaqaynayn alaabada firfircoon ama wax laga beddelay).
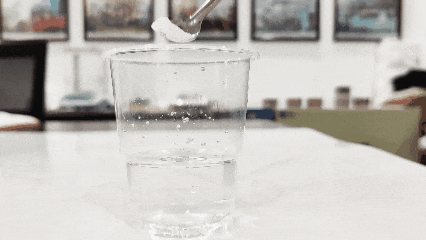

(4)Hubi inuu ku milmi karo biyaha. Guud ahaan, titanium dioxide waxaa lagu milmaa biyaha (marka laga reebo titanium dioxide si gaar ah loogu talagalay balaastikada, khad, iyo qaar ka mid ah titanium dioxide-ka synthetic-ga ah, kuwaas oo aan ku milmi karin biyaha).

II. Hababka kiimikada:
(1) Haddii budada kaalshiyamka lagu daro: Ku darista aashitada hydrochloric waxay sababi doontaa falcelin xooggan oo leh dhawaaq qaylo leh, oo ay weheliso soo saarista tiro badan oo xumbo ah (sababtoo ah kaalshiyamka kaarboonat wuxuu la falgalaa aashitada si uu u soo saaro kaarboon laba ogsaydh).

(2) Haddii lithopone lagu daro: Ku darista aashitada sulfuric-ga khafiifka ah ama aashitada hydrochloric waxay soo saari doontaa ur ukun qudhuntay.

(3) Haddii muunaddu ay tahay mid hydrophobic ah, ku darista hydrochloric acid ma sababi doonto falcelin. Si kastaba ha ahaatee, ka dib marka lagu qooyo ethanol ka dibna lagu daro hydrochloric acid, haddii xumbo la soo saaro, waxay caddaynaysaa in muunaddu ay ku jirto budada calcium carbonate ee dahaarka leh.

III. Waxaa sidoo kale jira laba hab oo kale oo wanaagsan:
(1) Marka la isticmaalo qaacidada isku midka ah ee PP + 30% GF + 5% PP-G-MAH + 0.5% budada titanium dioxide, xoogga yar ee walaxda ka dhalatay ayaa sii kordhaya tayada titanium dioxide (rutile) ee dhabta ah.
(2) Dooro resin hufan, sida ABS hufan oo lagu daray budada titanium dioxide 0.5%. Cabbir gudbinta iftiinka. Inta ay hoos u dhacdo gudbinta iftiinka, ayaa budada titanium dioxide-ka ay sii saxsan tahay.
Waqtiga boostada: Maajo-31-2024





